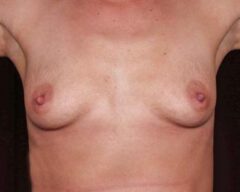
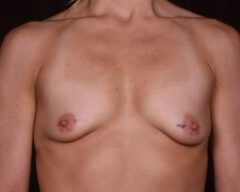
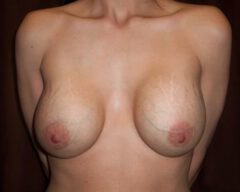
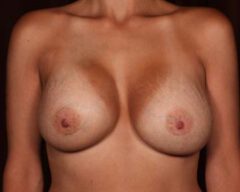
Changes in Aesthetic Preference
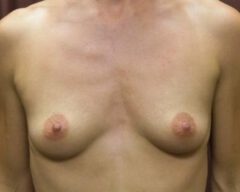
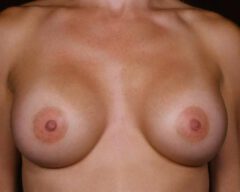
Our bodies evolve over time, and so do our preferences. A woman who wanted fuller breasts in her 20s may feel differently in her 40s or 50s. Some patients find that their implants no longer suit their frame, their wardrobe or their lifestyle. Others simply prefer a more natural silhouette. Whatever the reason, choosing to remove your implants because they no longer align with your vision for yourself is completely valid.
Rupture, Capsular Contracture & Other Complications
While the vast majority of women enjoy their implants for years without issue, complications can occur. Implant rupture, whether from trauma or gradual wear, may require removal or replacement. Saline implants deflate noticeably when ruptured, while silicone ruptures can be more subtle and may require imaging to detect.
Capsular contracture is another common reason for removal. This occurs when the scar tissue that naturally forms around an implant begins to tighten and harden, causing discomfort, distortion or both. Other complications that may prompt removal include implant displacement, asymmetry, rippling and infection.
Implant Age
Breast implants can last for 20 years or more, but are not meant to be lifetime devices. As they age, the risk of issues such as leakage or rupture becomes more likely. While the removal of intact implants isn’t necessary regardless of when they were placed, some women choose to have them removed or exchanged due to concerns about implant durability or integrity.
Health Concerns
Breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) is a rare type of cancer that has been linked to textured implants. BIA-ALCL is not breast cancer but a type of lymphoma that forms in the capsular tissue surrounding implants, typically presenting as swelling or fluid accumulation. If BIA-ALCL is confirmed with fluid testing and caught early, surgical removal of the implants and surrounding capsule is often the only treatment needed. According to the American Society of Plastic Surgeons (ASPS), only 1,618 cases have been reported worldwide, and there are no confirmed cases in patients with smooth implants.