Before the Procedure
You will have an ultrasound imaging exam of the leg that is to be treated. This exam is important for assessing the diseased superficial vein and planning the procedure.

The VenaSeal™ closure system is designed to close varicose veins by injecting a very small amount of proprietary medical adhesive (glue) inside of the vein. It is indicated for use in the permanent closure of lower extremity superficial truncal veins, such as the great saphenous vein (GSV), through endovascular embolization with coaptation. The vein is then compressed with ultrasound and remains closed.

Venaseal is the only FDA approved procedure that uses injection of medical adhesive to close veins. Because this adhesive works immediately to cause venous closure, compression stockings do not need to be worn after the procedure. Thermal procedures such as RFA and EVLA or chemical irritation of the vein, do not cause immediate closure, so compression stockings need to be worn post procedure.
VenaSeal is intended for use in adults with clinically symptomatic venous reflux as diagnosed by duplex ultrasound (DUS). VenaSeal system is contraindicated when any of the following conditions exist: previous hypersensitivity reactions to the VenaSeal adhesive or cyanoacrylates, acute superficial thrombophlebitis, thrombophlebitis migrans and acute sepsis.
Although this procedure can be used on many patients with superficial venous reflux, many insurance companies do not cover this procedure. We currently only perform this procedure on patients with eligible insurance coverage. Patients, who wish to pay for this procedure out of pocket, should contact the office for a quote.

You will have an ultrasound imaging exam of the leg that is to be treated. This exam is important for assessing the diseased superficial vein and planning the procedure.

The provider can discuss the procedure with you. It is performed as an outpatient in the office. The procedure typically takes 1-2 hours and only requires a small amount of local anesthetic (numbing medication). A brief summary of what to expect is below:

The provider will recommend follow-up care as needed. Compression stockings do not need to be worn after a Venaseal procedure. There is a follow-up ultrasound in 2 weeks to verify that the vein remained closed. Additional follow-up office appointments with ultrasound will be performed at 3 months and 12 months following the procedure in order to document long term closure of the treated vein.
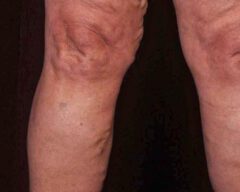
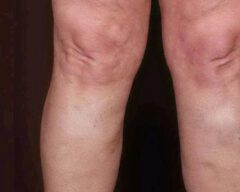
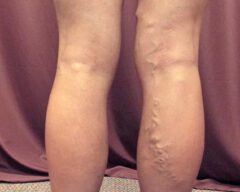
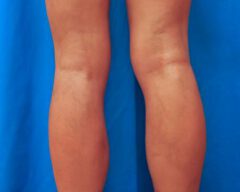
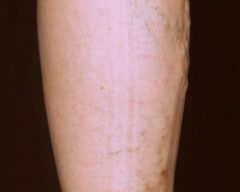
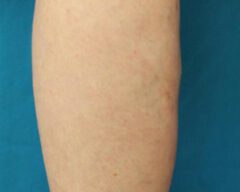
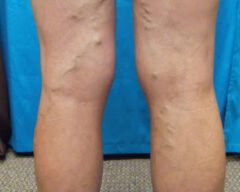
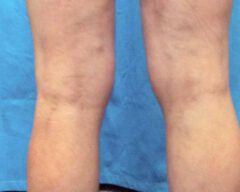
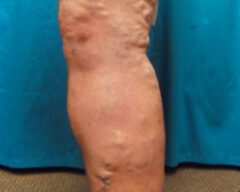
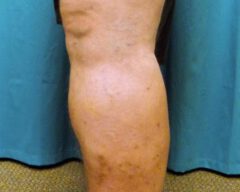
Before & Afters

While many insurance carriers cover varicose vein treatment, some individual insurance companies may limit the types of therapy which are covered. Coverage may depend on the severity of the varicose veins and symptoms. We will check to see if your plan covers this expensive, but safer treatment. Our experience with insurance pre-authorization showed limited insurance coverage for this treatment. Coverage changes routinely so pre-authorization is required. Patients are still ultimately responsible for any co-pays, deductibles, and any other non-covered insurance charges.
Patients without insurance or who do not wish to use insurance, should call for an individual consultation to get a custom quote for vein treatment.
Symptoms are caused by the diseased superficial vein. Thus, symptoms may improve as soon as the diseased vein is closed.
The VenaSeal procedure is designed to reduce recovery time. After the procedure patients should walk for 10-15 minutes. Over the next two weeks, walk daily and avoid very long periods of inactivity. Depending on the symptoms many patients are able to return to normal activities very shortly following the procedure.
To learn more about this procedure, contact the Vein Center at Fox Valley Plastic Surgery at 920-233-1540 to request a personal vein consultation.
Most patients feel little, if any, pain during the outpatient procedure. Most patients report little-to-no bruising after the VenaSeal procedure.
Venaseal is FDA approved for the treatment of superficial venous insufficiency/reflux. All medical procedures have some risk. The adverse events associated with the device are similar to those with traditional endovenous thermal ablation procedures. In addition, there are several risks unique to the VenaSeal system due to its material and product design as an implant.
These potential adverse events include, but are not limited to, allergic reactions to cyanoacrylates, such as hives, asthma, hay fever and anaphylactic shock, arteriovenous fistula, bleeding from the site of access, deep vein thrombosis (DVT), edema in the treated leg, embolization, including pulmonary embolism (PE), hematoma, hyperpigmentation, infection at the access site, non-specific mild inflammation of the cutaneous and subcutaneous tissue, pain, paresthesia, phlebitis, superficial thrombophlebitis, urticaria or ulceration may occur at the site of injection, vascular rupture and perforation, visible scarring.
Only a very small amount of VenaSeal adhesive is used to close the vein. The body will naturally create scar tissue around the adhesive over time to keep the vessel permanently closed. This adhesive is considered a medical device and will be left in the vein.
We recommend the purchase of Dermaka Cream for $34 to minimize bruising and inflammatory responses of the skin after a procedure. It works best by applying it five days before a procedure, and then after the procedure until no longer needed. Our patients have had excellent results with the cream, which can also be used for eczema, rashes, dry skin, etc. Learn more on Dermaka.com, or download the patient booklet here.
Take the first step toward renewed confidence with a consultation at Fox Valley Plastic Surgery. Our team is here to listen, guide, and help you achieve results that feel true to you.